Developing
Adoptive
Cell Therapies
Immatics’ clinical ACTengine® programs are based on genetically engineering a patient’s own, autologous T cells with novel TCRs designed to recognize the cancer target on the tumor. The engineered T cells (TCR-T) aim to induce a robust and specific anti-tumor attack to fight the cancer.
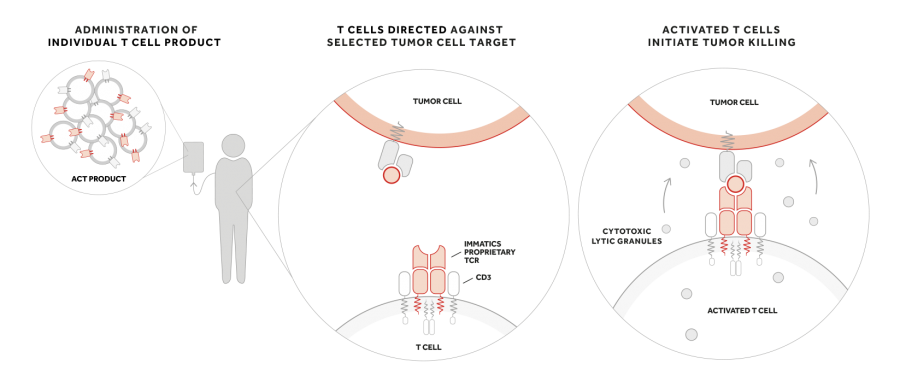
Upon infusion of an ACTengine® product, T cells “equipped” with the cancer target-specific TCR are supposed to bind to the specific target on the tumor. Subsequent activation of the T cell induces release of cytotoxic granules that might ultimately lead to tumor killing.
ACTengine®
at a glance
Immatics’ clinical product class ACTengine® is a personalized approach in which the patient’s own T cells (autologous) are genetically modified to express a novel proprietary TCR, an approach also known as TCR-T (T cell receptor-engineered T cell). The modified T cells are then reinfused into the patient to specifically engage with the tumor. The product class ACTallo® is advancing the ACT concept beyond individualized manufacturing and is being developed to generate a next-generation of cell therapies, our allogeneic “off-the-shelf” cell therapies.
Expanded target space compared to CAR-T
ACTengine® TCR-T product candidates target tumor-associated peptides presented by HLA-molecules on the tumor cell surface. Most relevant solid cancer targets are of intracellular nature and can only be accessible by TCR-based approaches.
TCRs with desirable affinity and specificity
ACTengine® TCRs identified via the XCEPTOR® TCR discovery platform and characterized via XPRESIDENT® guided on- and off-target toxicity screening show desirable affinity and high specificity for their cancer cell target. Immatics’ competitive advantage to other TCR-T approaches is our ability to combine the most suitable target (via XPRESIDENT®) with the right TCR (via XCEPTOR®).
Active at physiological levels of target expression
Immatics believes that ACTengine® TCR-T product candidates are highly potent and capable of inducing the killing of tumor cells that present the target at physiological target copy numbers identified by quantitative mass spectrometry and TCR validation.
Optimized manufacturing
Immatics’ proprietary short manufacturing process, which includes a short manufacturing period and a proprietary cytokine cocktail used to promote T cell expansion in cell culture, is designed to produce younger, less differentiated T cell phenotypes that are associated with better T cell engraftment and persistencein the patient’s blood.
Clinical trials
The objective of the Phase 1 clinical trials with the ACTengine® product candidates, IMA203 and IMA203CD8 is to evaluate safety, tolerability and initial signs of clinical and biological efficacy in target-positive solid cancer patients. The clinical trials continue to actively recruit patients.
ACTengine® IMA203 TARGETING PRAME
Based on interim results from the Phase 1 dose escalation, we expanded the IMA203 trial to three cohorts: IMA203 as monotherapy, IMA203 in combination with an immune checkpoint inhibitor and IMA203CD8, a 2nd generation monotherapy where IMA203 is co-transduced with a CD8 co-receptor to leverage the both, CD8 and CD4 T cells, to fight the tumor.
There are several reasons why a patient’s own T cells are often not able to protect the body against cancer. These include the unavailability of activated tumor antigen specific T cells or the insufficient affinity of endogenous target-specific TCRs to properly activate the T cell and destroy the tumor. Immatics’ approach to solving these problems is to engineer autologous T cells with a well characterized and potent TCR, which is the underlying principle of its ACTengine® approach.
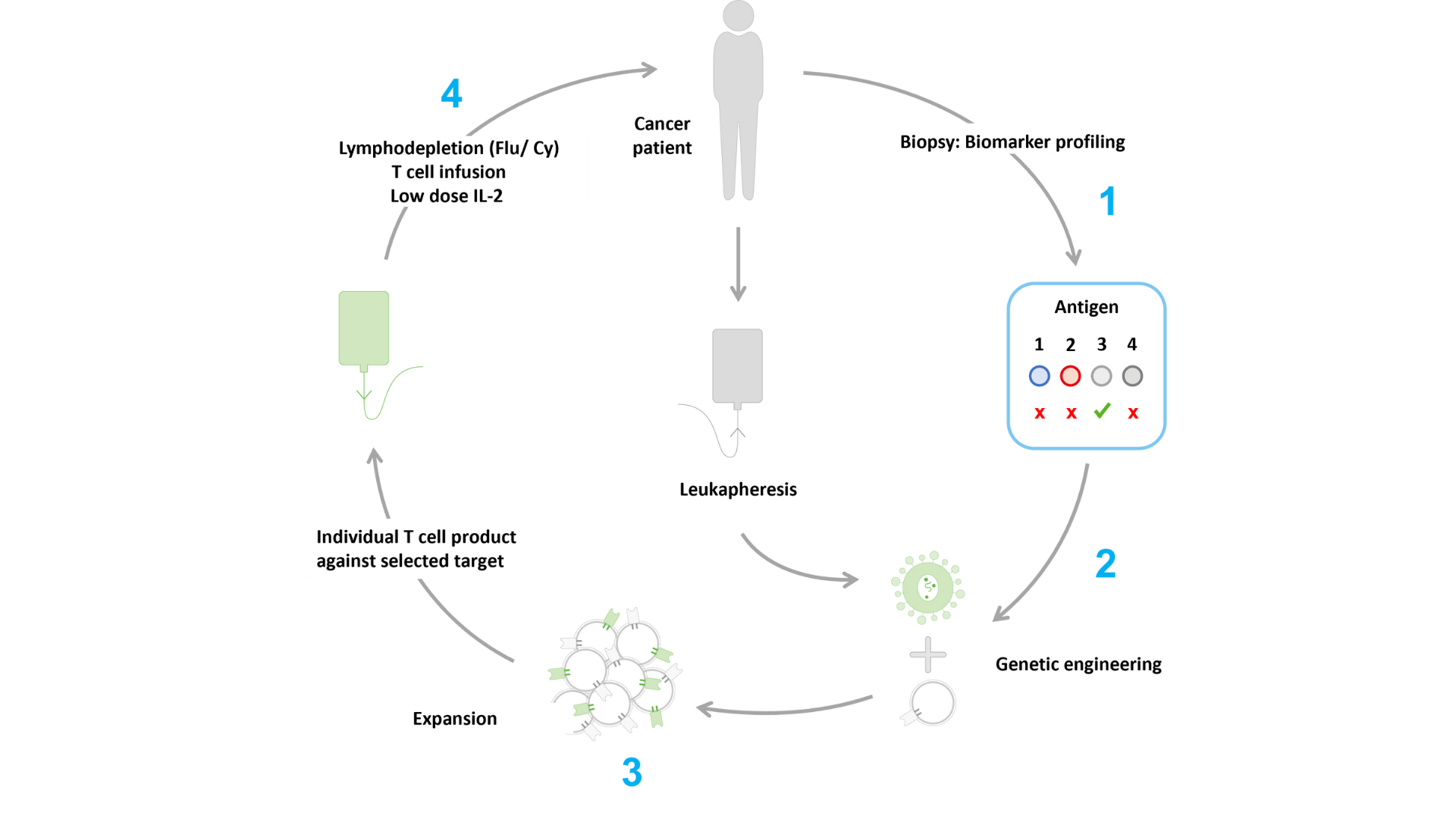
ACTengine® is based on genetically engineering a patient’s own T cells with a novel TCR designed to recognize the cancer target that is identified by Immatics’ target discovery platform XPRESIDENT®. (1) The target of interest is first confirmed on a patient’s tumor by Immatics’ proprietary IMADetect® companion diagnostic device candidate. (2) By lentiviral transduction, a patient’s autologous T cells are equipped with a target-specific TCR developed by Immatics. (3) The engineered T cells are then expanded in vitro and the ACTengine® product is ready for infusion into the patient. (4) The patient will receive a pre-treatment, called lymphodepletion, which aims to prepare the patient’s body for the infusion of ACTengine® T cells. By temporarily removing immune cells in the patient’s blood we aim to make room for the engineered T cells and help them to engraft and expand to numbers large enough to fight the tumor. Following infusion, a low dose of IL-2, an immune system signaling molecule, is administered for 14 days to further enhance persistence of the infused engineered T cells.

